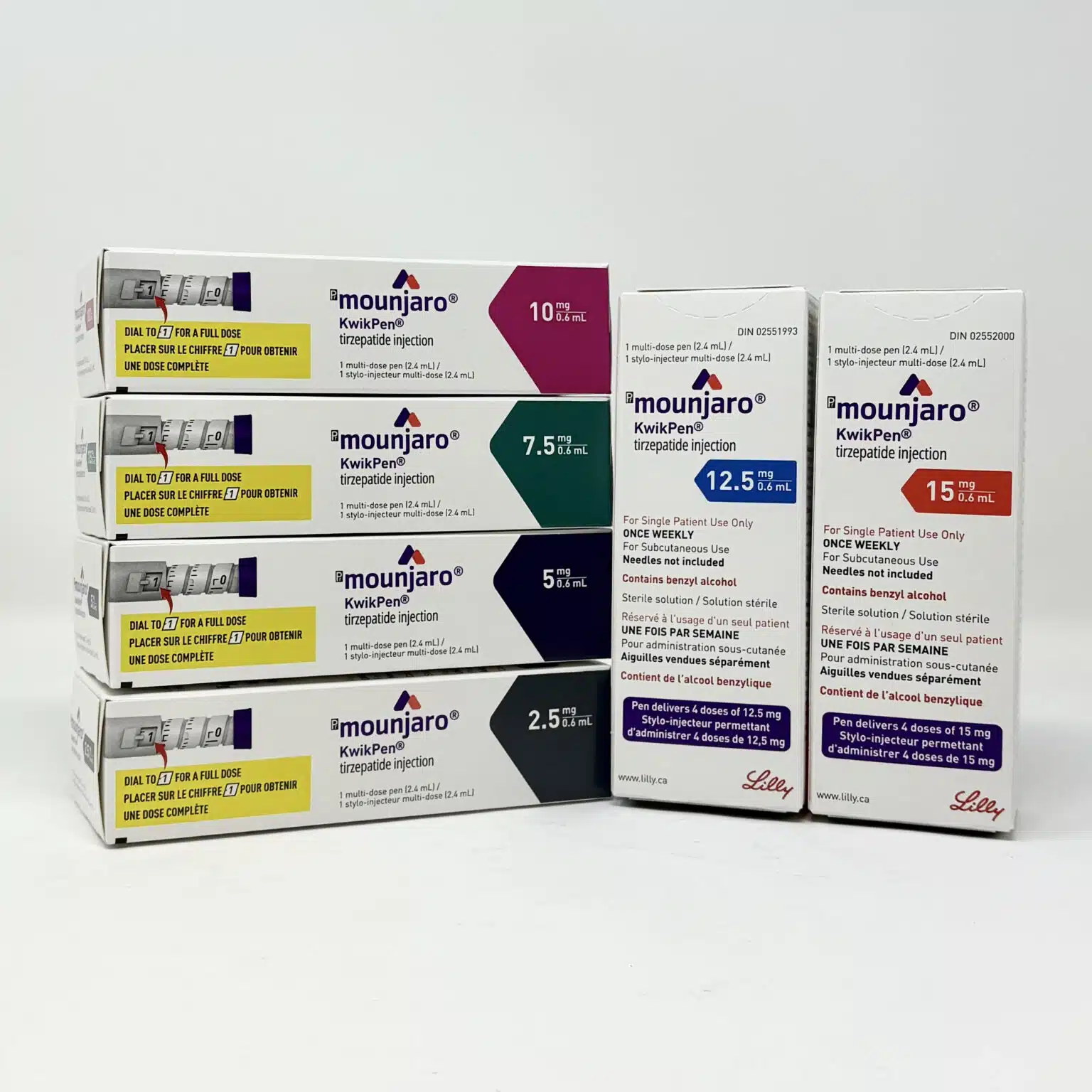
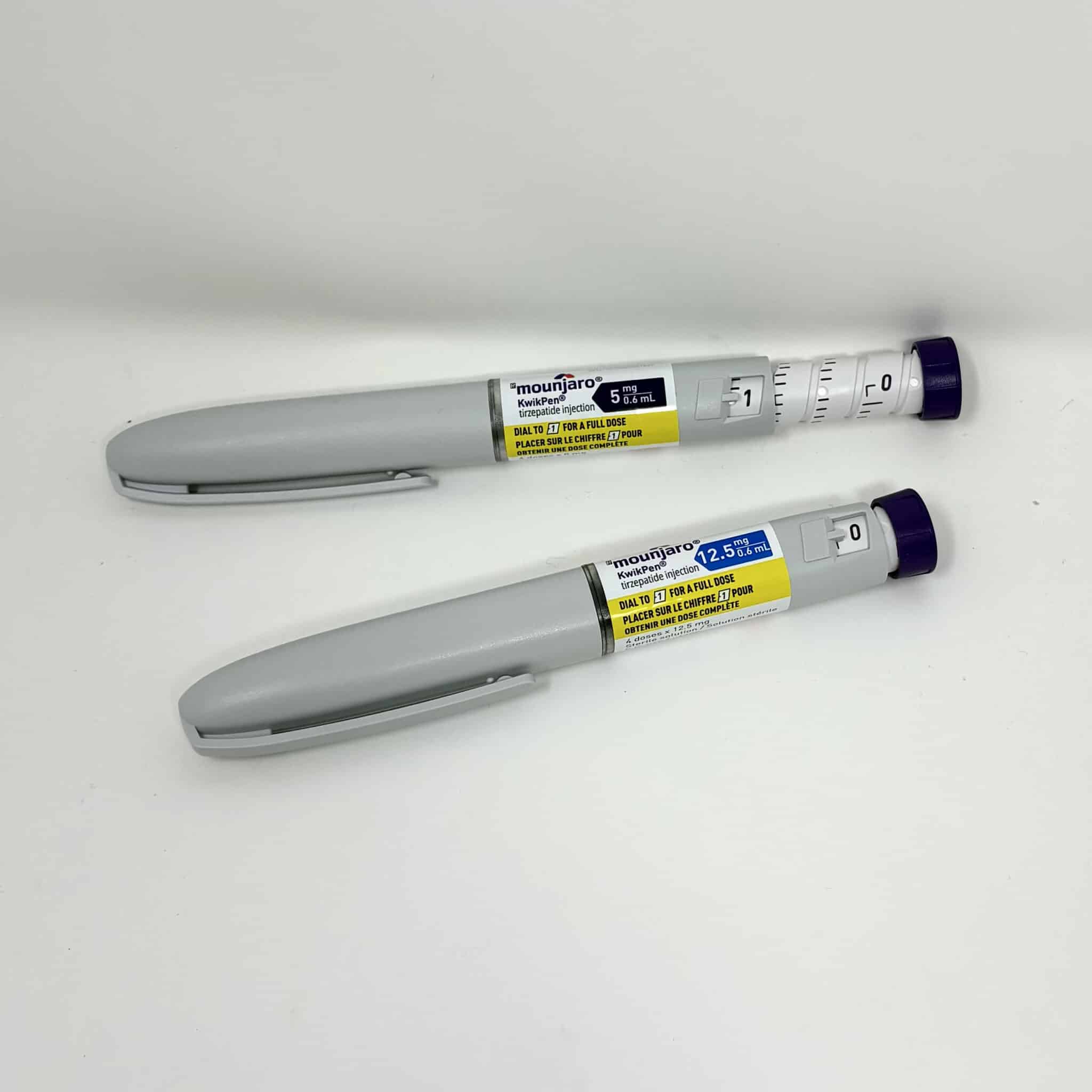
Mounjaro® KwikPen (Tirzepatide) Injection
Tirzepatide is the active ingredient in Mounjaro®, a once-weekly injectable treatment developed by Eli Lilly for adults with type 2 diabetes mellitus. The KwikPen® presentation provides a pre-filled, multi-dose device designed to improve dosing accuracy and treatment adherence.
Each KwikPen contains four doses (a 4-week supply). New customers may receive 10% off their first order using coupon code KWIKPEN, subject to eligibility.
What Is Mounjaro KwikPen?
Mounjaro KwikPen is a prefilled injection pen containing tirzepatide, a first-in-class dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. It is indicated for improving glycaemic control in adults with type 2 diabetes, alongside diet and exercise.
From January 2025, the KwikPen format is expected to replace vial presentations, aligning with the manufacturer’s transition plan. Patients currently using vials can learn more via our Mounjaro vial information page.
How Mounjaro Works
Tirzepatide uniquely activates both GIP and GLP-1 receptors. This dual incretin effect:
- Enhances glucose-dependent insulin secretion
- Reduces glucagon levels when glucose is elevated
- Improves insulin sensitivity
- Delays gastric emptying
- Reduces appetite
Together, these mechanisms support improved HbA1c reduction and may contribute to clinically meaningful weight loss in appropriate patients.
Why Choose the KwikPen Format?
- Pre-filled convenience: No manual drawing from vials.
- Four weekly doses per pen: Supports consistent adherence.
- Accurate dosing: Engineered for precise subcutaneous administration.
- Improved usability: Designed for patient-friendly self-injection.
- Transition from vials: Vials are being phased out by early 2025.
Mounjaro vs Semaglutide
Semaglutide is marketed under brand names such as Ozempic® and Wegovy®. While both medicines are incretin-based therapies, key differences exist:
Mechanism of Action
Mounjaro acts on both GIP and GLP-1 receptors, whereas semaglutide targets only GLP-1 receptors.
Clinical Effectiveness
Comparative clinical trial data suggest tirzepatide may produce greater average reductions in HbA1c and body weight than semaglutide, though individual responses vary.
Dosing Range
Mounjaro doses range from 2.5 mg to 15 mg once weekly. Semaglutide dosing differs depending on indication.
Side Effects
Both medicines commonly cause gastrointestinal effects such as nausea and diarrhoea. Tolerability varies between individuals.
Dosage and Administration
Mounjaro KwikPen is administered once weekly via subcutaneous injection.
- Starting dose: 2.5 mg weekly for 4 weeks
- Increase to: 5 mg weekly
- Further titration: Increase in 2.5 mg increments every 4 weeks as needed
- Maximum dose: 15 mg weekly
Injection Guidance
- Inject into the abdomen, thigh or upper arm.
- Rotate sites weekly to reduce irritation.
- Avoid intramuscular or intravenous administration.
Always follow your prescriber’s instructions regarding dose adjustments.
Mounjaro Dosage and Average Weight Loss
| Weekly Dose (mg) | Average Weight Loss (kg) | Average Weight Loss (lbs) |
|---|---|---|
| 2.5 mg | 10.1 kg | 22.3 lbs |
| 5 mg | 12.6 kg | 27.8 lbs |
| 10 mg | 19.5 kg | 42.9 lbs |
| 15 mg | 22.6 kg | 49.8 lbs |
These figures demonstrate a correlation between higher weekly doses and greater average weight reduction in clinical trials. Dose selection must balance efficacy with tolerability.
Storage and Handling
- Refrigerate at 2–8°C until expiry.
- Do not freeze.
- May be stored below 30°C for up to 30 days if required.
- Keep in original packaging to protect from light.
Dispose of used pens in a puncture-resistant sharps container. Never share injection devices.
Side Effects
Common
- Nausea
- Vomiting
- Diarrhoea
- Reduced appetite
- Abdominal discomfort
Serious (Rare)
- Pancreatitis
- Severe hypersensitivity reactions
- Thyroid tumours (observed in animal studies)
Seek urgent medical attention if severe abdominal pain, persistent vomiting or breathing difficulties occur.
Special Considerations
Pregnancy
Mounjaro is contraindicated during pregnancy. Effective contraception is recommended, and treatment should be discontinued at least one month before planned conception.
Oral Contraceptives
Due to delayed gastric emptying, tirzepatide may reduce absorption of oral contraceptives during initiation. Additional barrier contraception is recommended for at least four weeks after starting therapy.
Drug Interactions
Delayed gastric emptying may alter absorption of certain oral medicines, including antibiotics and antiplatelet agents. Discuss all current treatments with your healthcare provider.
Switching from Vials to KwikPen
- Confirm your prescribed dose matches the KwikPen format.
- Review administration instructions before first use.
- Consult your pharmacist or clinician if unsure about technique.
Buying Mounjaro KwikPen from Canada
Canadian pharmacies operate under regulated pricing frameworks, often resulting in lower medication costs compared to U.S. retail pricing. Patients with a valid prescription may order through reputable Canadian prescription referral services.
- Obtain a valid prescription.
- Select a licensed pharmacy provider.
- Submit prescription and shipping details.
- Receive temperature-controlled delivery.
Frequently Asked Questions
What is Mounjaro KwikPen used for?
It is prescribed to improve blood glucose control in adults with type 2 diabetes.
Is Mounjaro effective for weight loss?
Clinical trials show significant average weight reduction alongside glycaemic improvement.
What is the starting dose?
2.5 mg once weekly for four weeks, then increased according to response.
Can the pen be micro-dosed?
Some patients enquire about click-counting strategies. For detailed information, see Microdosing Mounjaro KwikPen guidance.