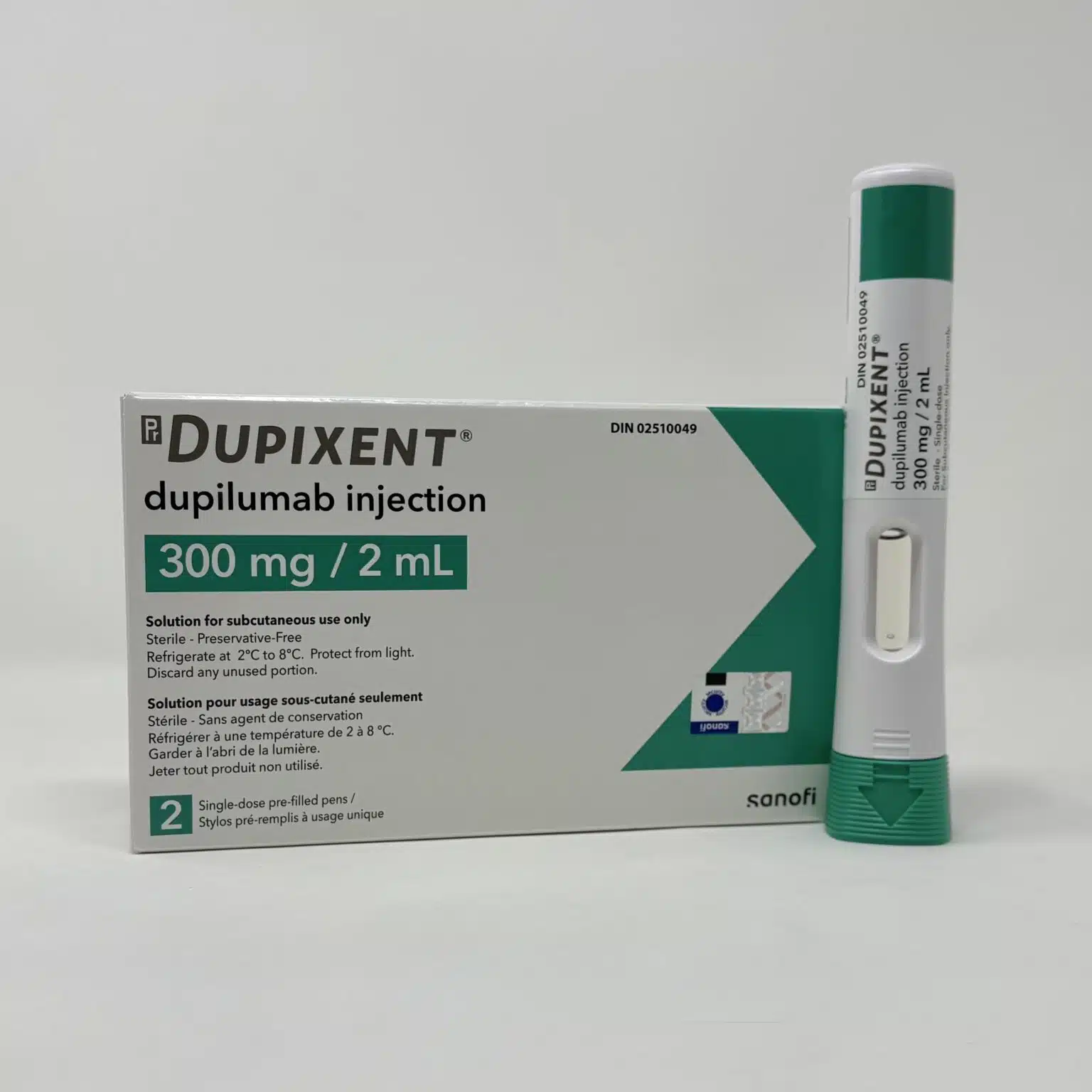
Dupixent® (Dupilumab) Injection
Dupixent® (dupilumab) is a targeted biologic therapy indicated for multiple chronic inflammatory diseases driven by type 2 inflammation, including atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyps, eosinophilic oesophagitis, prurigo nodularis, chronic spontaneous urticaria, and selected cases of COPD with raised eosinophils. Administered as a subcutaneous injection using a pre-filled pen or syringe, Dupixent is designed for long-term disease control under specialist supervision.
What Is Dupixent?
Dupixent contains dupilumab, a fully human monoclonal antibody that targets key immune pathways involved in type 2 inflammatory responses. It is authorised for use in adults, adolescents and, for certain indications, children meeting defined age and weight criteria.
Type 2 inflammation plays a central role in conditions such as eczema (atopic dermatitis), eosinophilic asthma and chronic rhinosinusitis with nasal polyps. By selectively inhibiting specific interleukin signalling pathways, Dupixent reduces inflammatory activity without broadly suppressing the immune system.
Regulatory approval has been granted following extensive clinical evaluation by authorities including the European Medicines Agency (EMA) and national regulatory bodies. Treatment decisions in the UK are guided by specialist clinicians in accordance with evidence-based recommendations from the National Institute for Health and Care Excellence (NICE).
Therapeutic Indications
Dupixent may be prescribed as add-on therapy or maintenance treatment for the following conditions, depending on clinical criteria:
- Atopic dermatitis (eczema) – moderate to severe disease not adequately controlled with topical prescription therapies.
- Asthma – including severe eosinophilic asthma and asthma requiring maintenance oral corticosteroids in selected patients aged 12 years and older.
- Chronic obstructive pulmonary disease (COPD) in adults with raised eosinophil levels, as add-on maintenance therapy.
- Chronic rhinosinusitis with nasal polyposis (CRSwNP) – as add-on therapy with intranasal corticosteroids in severe disease inadequately controlled by systemic steroids and/or surgery.
- Eosinophilic oesophagitis (EoE) – in patients aged 1 year and above weighing at least 15 kg.
- Prurigo nodularis (PN) – moderate to severe disease not adequately controlled with prescription topical therapy.
- Chronic spontaneous urticaria (CSU) – in patients aged 12 years and older whose symptoms are not adequately controlled with H1 antihistamines.
The appropriateness of treatment is determined by a healthcare professional based on disease severity, previous therapy response and patient-specific risk factors.
Mechanism of Action: How Dupixent Works
Dupilumab selectively blocks the shared receptor component for interleukin-4 (IL-4) and interleukin-13 (IL-13). These cytokines are central drivers of type 2 inflammation.
By inhibiting IL-4 and IL-13 signalling, Dupixent reduces:
- Eosinophilic airway inflammation in asthma and COPD with eosinophilia
- Skin barrier dysfunction and inflammatory lesions in atopic dermatitis
- Nasal polyp growth and mucosal inflammation in CRSwNP
- Oesophageal inflammation in eosinophilic oesophagitis
- Chronic itch and inflammatory nodules in prurigo nodularis
Unlike traditional systemic immunosuppressants, dupilumab specifically targets defined cytokine pathways rather than broadly suppressing immune activity.
Available Forms and Strengths
Dupixent is supplied as a sterile solution for subcutaneous injection in the following presentations:
- 300 mg/2 mL single-use pre-filled syringe or pre-filled pen
- 200 mg/1.14 mL single-use pre-filled syringe (with needle shield) or pre-filled pen
The choice of strength and dosing interval depends on the treated condition, patient age and, in paediatric cases, body weight.
Dosing Overview
Dosing schedules vary by indication. Some adult regimens begin with a loading dose (two injections on the first day), followed by maintenance dosing.
Examples of Adult Dosing
- COPD: 300 mg every two weeks.
- CRSwNP: 300 mg every two weeks.
- Prurigo nodularis: 600 mg loading dose, then 300 mg every two weeks.
- Chronic spontaneous urticaria: 600 mg loading dose, then 300 mg every two weeks.
- Asthma (selected patients ≥12 years requiring oral corticosteroids): 600 mg loading dose, then 300 mg every two weeks.
- Eosinophilic oesophagitis: Weight-based dosing; some patients receive 300 mg weekly, while others receive 300 mg every two weeks depending on weight.
Paediatric dosing is weight-dependent and indication-specific. Always follow the prescribing clinician’s instructions precisely.
Missed Dose Advice
- Weekly schedule: Administer the missed dose as soon as possible and begin a new weekly schedule from that date.
- Every two weeks: If within 7 days of the missed dose, inject as soon as possible and resume the original schedule. If more than 7 days have passed, skip the missed dose and wait for the next scheduled date.
How to Inject Dupixent Safely
Dupixent is administered via subcutaneous injection only. It must not be injected intravenously or intramuscularly.
Pre-Injection Checks
- Confirm the correct dose and product.
- Check the expiry date.
- Inspect the solution; it should appear clear and colourless to pale yellow.
- Do not use if cloudy, discoloured or containing visible particles.
- Do not shake, freeze or expose to heat.
Allow to Reach Room Temperature
- 300 mg presentations typically require approximately 45 minutes at room temperature.
- Some 200 mg presentations require approximately 30 minutes.
- Do not accelerate warming using artificial heat sources.
Injection Sites
- Front of thigh
- Abdomen (avoid 5 cm around the navel)
- Outer upper arm (if administered by a caregiver)
- Rotate sites for each injection
- For loading doses requiring two injections, separate sites by at least 5 cm
Pen and Syringe Guidance
Follow the device-specific “Instructions for Use” supplied in the carton. Do not re-use pens or syringes. Dispose of all used devices in a puncture-resistant sharps container in accordance with local regulations.
Common Side Effects
- Injection site reactions
- Conjunctivitis and other eye-related inflammation (more common in atopic dermatitis)
- Upper respiratory symptoms
- Herpes virus infections in some patient groups
Patients should promptly report new or worsening eye symptoms such as redness, pain, blurred vision or discharge.
Serious Adverse Reactions
Seek urgent medical attention if you experience:
- Signs of severe allergic reaction (angioedema, breathing difficulty, hives)
- Severe rash or systemic hypersensitivity
- Symptoms suggestive of vasculitis or eosinophilic complications
Treatment should be discontinued immediately if a serious hypersensitivity reaction is suspected.
Warnings and Precautions
- Not for acute asthma attacks: It does not treat acute bronchospasm or status asthmaticus.
- Corticosteroid tapering: Do not abruptly discontinue systemic, inhaled or topical corticosteroids.
- Eosinophilic conditions: Monitor for signs of systemic eosinophilia, including pulmonary or vasculitic symptoms.
- Helminth infections: Treat pre-existing infections before initiation.
- Eye inflammation: Persistent symptoms warrant ophthalmologic evaluation.
- Arthralgia: Report new or worsening joint pain.
Storage and Handling
- Refrigerate at 2°C–8°C.
- Keep in original carton to protect from light.
- Do not freeze or shake.
- May be stored at room temperature (up to 25°C) for a maximum of 14 days.
- Discard if left out longer than 14 days.
Dupixent Compared with Other Treatments
Dupixent is not a corticosteroid and is not classified as a traditional systemic immunosuppressant. It may be used alongside:
- Topical corticosteroids or calcineurin inhibitors for eczema
- Inhaled maintenance therapies for asthma or COPD
- Intranasal corticosteroids for CRSwNP
Treatment selection depends on disease severity, previous therapeutic response and comorbidities.
Buying Dupixent Online
Biologic therapies can be costly, particularly for patients with high deductibles or limited coverage. LifePath Meds supports eligible patients seeking more predictable pricing for brand-name medications such as Dupixent.
When ordering online, ensure:
- You have a valid prescription specifying dose and schedule.
- The pharmacy is properly licensed and regulated.
- You understand storage and shipping conditions.
Learn more about medication access options at LifePath Meds.