Zepbound KwikPen (Tirzepatide) Injection for Weight Management
Zepbound KwikPen is a pre-filled, once-weekly injectable pen containing tirzepatide for chronic weight management. Each pen delivers four doses, offering straightforward administration and consistent weekly dosing for eligible adults.
What Is Zepbound (Tirzepatide)?
Zepbound contains tirzepatide, a dual GIP and GLP-1 receptor agonist indicated for chronic weight management in adults. It is also used in adults with type 2 diabetes mellitus. Treatment should always form part of a broader plan that includes a reduced-calorie diet and increased physical activity, in line with guidance from the
National Institute for Health and Care Excellence (NICE) and the
NHS.
The active ingredient, tirzepatide, works on two incretin hormone pathways to help regulate appetite and glucose levels.
How Tirzepatide Works
Tirzepatide is a dual agonist of the glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) receptors. These hormones are involved in:
- Stimulating insulin secretion in a glucose-dependent manner
- Reducing appetite and food intake
- Slowing gastric emptying
- Supporting improved glycaemic control
By targeting both pathways, Zepbound helps decrease caloric intake and enhance insulin sensitivity. The delay in gastric emptying may also reduce post-meal glucose spikes.
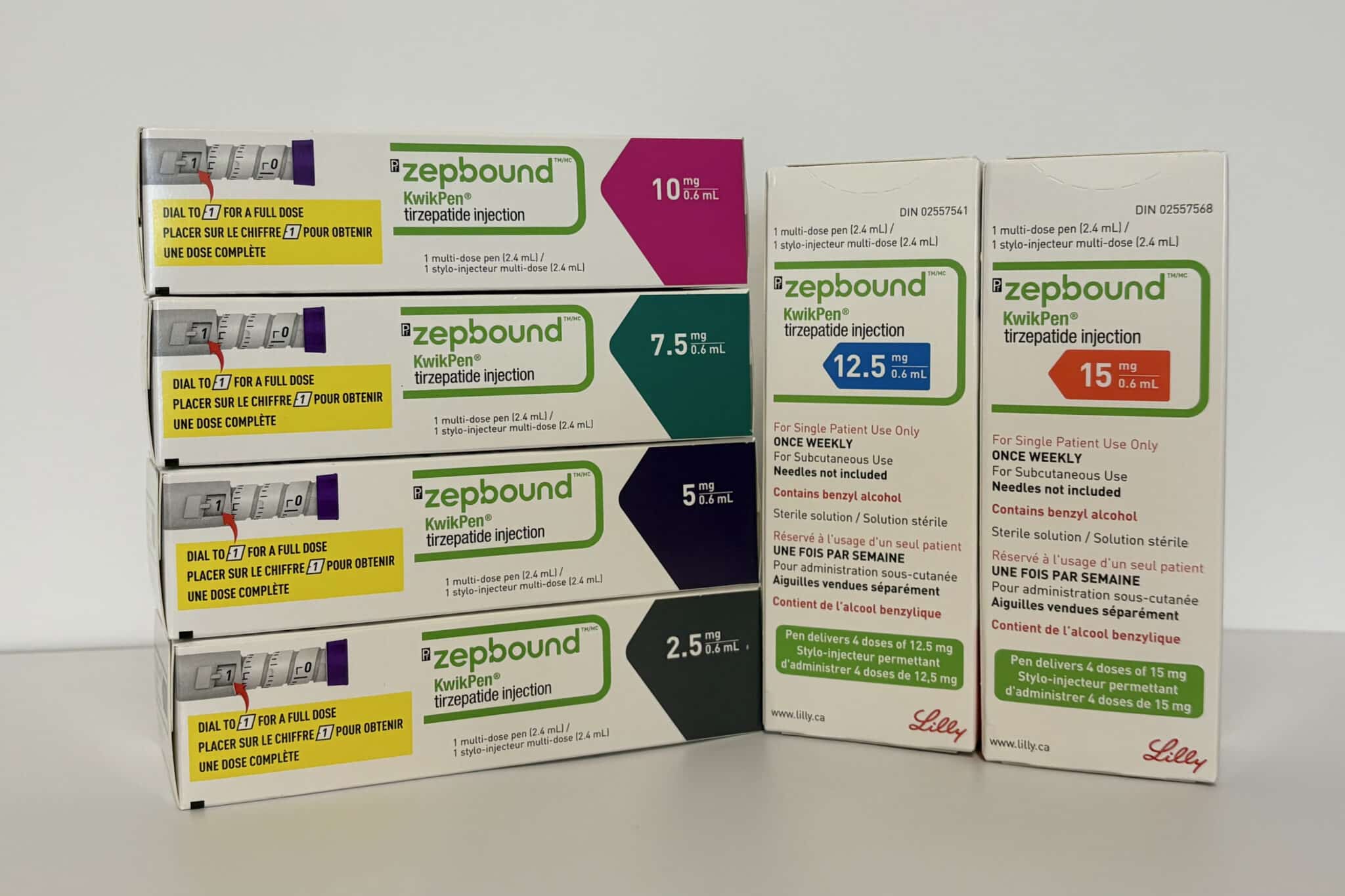
Available Strengths
Zepbound KwikPen is available in the following strengths:
- 2.5 mg
- 5 mg
- 7.5 mg
- 10 mg
- 12.5 mg
- 15 mg
Each pen contains four once-weekly doses.
Dosage and Administration
Starting and Titration Schedule
- Initial dose: 2.5 mg injected subcutaneously once weekly for 4 weeks.
- After 4 weeks: Increase to 5 mg once weekly.
- Further increases: If needed and tolerated, increase in 2.5 mg increments every 4 weeks.
- Maximum dose: 15 mg once weekly.
Injection Sites
Administer subcutaneously into the abdomen, thigh, or upper arm. Rotate injection sites with each dose to minimise local reactions.
Missed Dose Guidance
If a dose is missed, administer it within 4 days where possible. If more than 4 days have passed, skip the missed dose and resume the regular schedule.
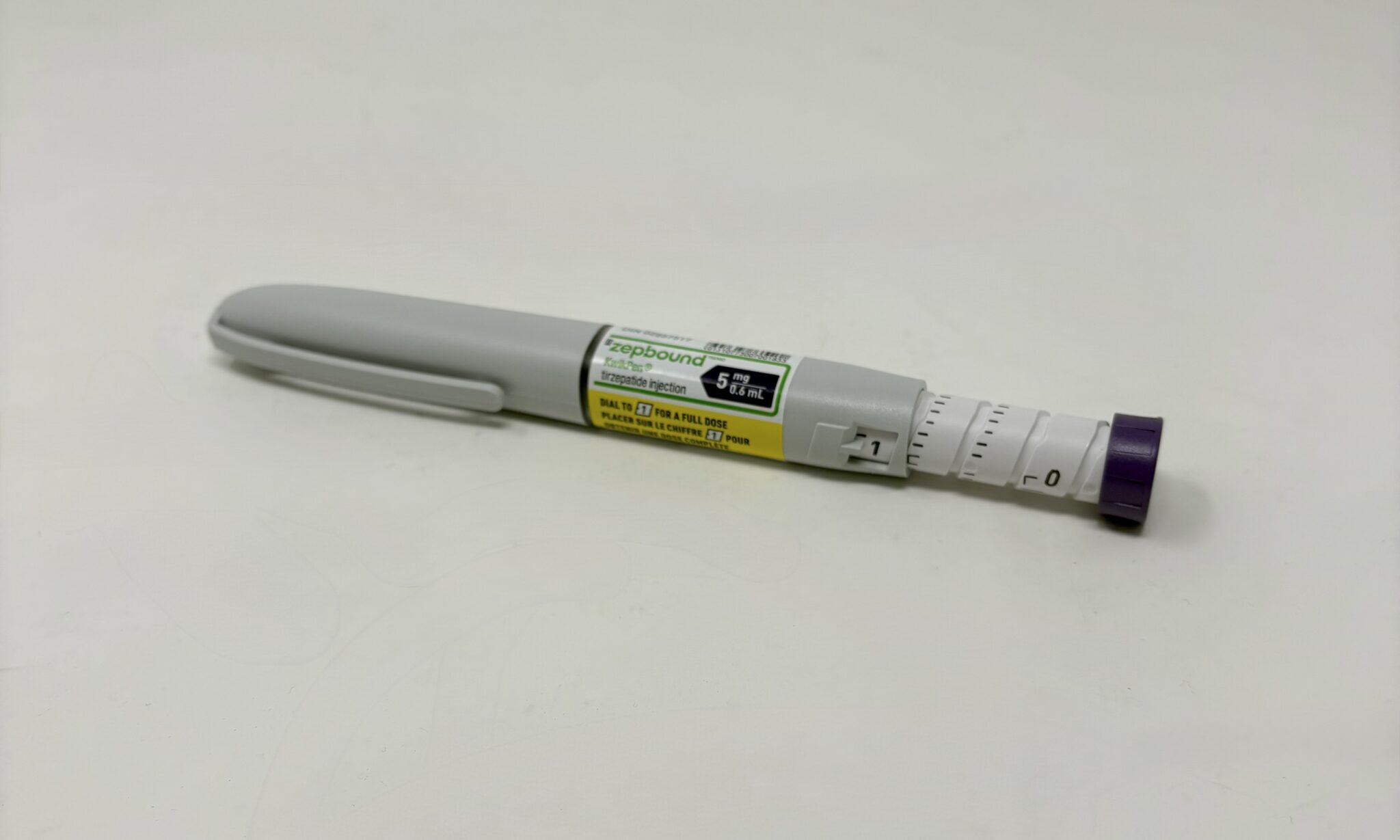
Step-by-Step Use
Before injecting, confirm the solution is clear and colourless. Attach a new needle, remove both caps, turn the dose selector to “1” regardless of prescribed strength, clean the injection site, insert the needle subcutaneously, press the injection button and hold until the counter returns to “0”.
Expected Weight Loss Outcomes
Clinical trials have demonstrated dose-dependent weight reduction over 72 weeks:
- 5 mg: Average 15% body weight reduction
- 10 mg: Average 19.5% reduction
- 15 mg: Average 20.9% reduction
Individual results vary based on adherence, lifestyle measures, and baseline characteristics.
Side Effects and Safety Information
Common Adverse Effects
- Nausea
- Diarrhoea
- Vomiting
- Constipation
- Abdominal discomfort
- Fatigue
- Hair loss
- Injection-site reactions
Serious Risks
- Thyroid C-cell tumours
- Pancreatitis
- Acute kidney injury
- Severe gastrointestinal complications
- Hypersensitivity reactions
- Gallbladder disease
- Suicidal ideation or behaviour
Seek urgent medical attention if you experience persistent abdominal pain, signs of pancreatitis, or symptoms of an allergic reaction.
Drug Interactions
Zepbound may interact with insulin and other glucose-lowering therapies. Due to delayed gastric emptying, absorption of oral medicines may be affected. Patients using oral contraceptives should consider non-oral or barrier contraception during initiation and dose escalation. Always disclose all medicines and supplements to your healthcare provider.
Precautions and Contraindications
- Not recommended in patients with a history of medullary thyroid carcinoma (MTC)
- Contraindicated in Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Use caution in patients with pancreatitis history
- Exercise care in severe gastrointestinal, kidney, or gallbladder disease
- Avoid during pregnancy
Consult your doctor if pregnant, planning pregnancy, or breastfeeding. Refer to the official prescribing information for comprehensive safety guidance.
Zepbound vs Mounjaro
Zepbound and
Mounjaro KwikPen contain the same active ingredient, tirzepatide, and are administered in identical dosing schedules. Zepbound is authorised for weight management, while Mounjaro is authorised for type 2 diabetes management.
Switching from Ozempic
Approximate dose comparisons when transitioning from Ozempic:
- Ozempic 0.25 mg ≈ Zepbound 2.5 mg
- Ozempic 0.5 mg ≈ Zepbound 5 mg or 7.5 mg
- Ozempic 1 mg ≈ Zepbound 10 mg
- Ozempic 2 mg ≈ Zepbound 12.5 mg or 15 mg
These are general guides only. Clinical supervision is required when switching therapies.
Buying Zepbound from Canada
Some U.S. patients obtain tirzepatide from licensed Canadian pharmacies due to pricing differences. A valid prescription is required. Ensure the pharmacy is properly regulated and that you consult your prescriber before importing medication.
Manufacturer coupons available in the United States generally do not apply to Canadian purchases.
Storage Instructions
- Refrigerate at 2°C to 8°C (36°F to 46°F)
- May be kept at room temperature (up to 30°C) for up to 21 days
- Do not freeze
- Store in original carton to protect from light
Frequently Asked Questions
What is Zepbound used for?
Zepbound is prescribed for long-term weight management in adults with obesity or overweight with weight-related conditions, alongside diet and exercise.
Is Zepbound available in Canada?
Zepbound is not yet available in Canadian pharmacies. Prescriptions may be dispensed as Mounjaro KwikPen, which contains the same active ingredient. Availability is expected in 2025.
How much weight can I lose?
Clinical trials report up to approximately 20% body weight reduction at the highest dose over 72 weeks, though results vary.
Where can I find official prescribing information?
Consult the manufacturer’s prescribing information and product monograph for detailed guidance.
Reviews
There are no reviews yet.