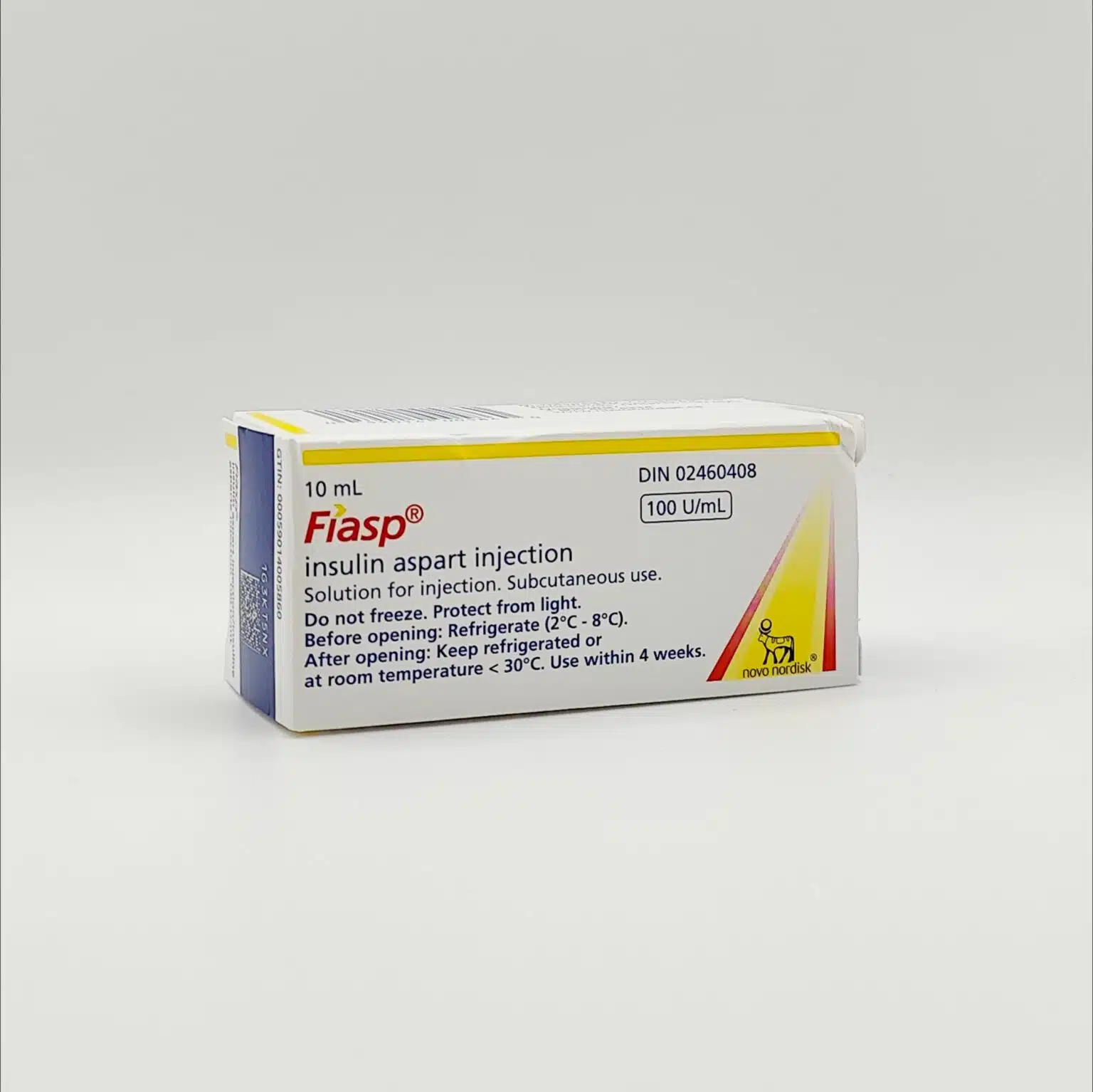
Fiasp (Insulin Aspart) 100 Units/mL Solution for Injection – 10 mL Vials
Rapid-acting mealtime insulin for adults and children with diabetes mellitus.
Overview of Fiasp Insulin Aspart
Fiasp is a fast-acting insulin aspart injection formulated to support glycaemic control in adults and paediatric patients diagnosed with Type 1 or Type 2 diabetes mellitus. Supplied in 10 mL multidose vials containing 100 units per millilitre (U/mL), Fiasp is administered subcutaneously around mealtimes to help regulate postprandial blood glucose levels. It may be prescribed as part of a comprehensive diabetes management plan that includes longer-acting (basal) insulin and, where appropriate, oral antidiabetic medicines.
Designed to mimic the body’s natural insulin response during food intake, Fiasp provides a more rapid onset of action compared with conventional insulin aspart formulations such as :contentReference[oaicite:0]{index=0}. This earlier activity profile supports improved management of post-meal glucose excursions when used according to medical advice.
What Is Fiasp Used For?
Fiasp (insulin aspart) is indicated for the treatment of diabetes mellitus in:
- Adults with Type 1 diabetes
- Adults with Type 2 diabetes requiring insulin therapy
- Children and adolescents with diabetes, as prescribed by a healthcare professional
Mealtime insulin plays a crucial role in limiting sharp rises in blood glucose following carbohydrate intake. Clinical guidance from the National Institute for Health and Care Excellence (NICE) guidelines on Type 1 diabetes and the NICE guidance on Type 2 diabetes management emphasises individualised insulin regimens to optimise glycaemic control while minimising hypoglycaemia risk.
How Fiasp Works: Mechanism of Action
The active ingredient, insulin aspart, is a rapid-acting analogue of human insulin. After subcutaneous injection, it facilitates the uptake of glucose from the bloodstream into muscle and adipose tissues and suppresses hepatic glucose production. This mechanism mirrors the physiological action of endogenous insulin.
Fiasp is formulated to begin lowering blood glucose approximately 2 minutes after injection, making it suitable for administration immediately before meals or shortly after eating begins. Its accelerated absorption profile distinguishes it from standard insulin aspart preparations, providing earlier postprandial glucose control.
According to information published by the European Medicines Agency (EMA) Fiasp assessment report, the formulation enables earlier insulin exposure compared with conventional insulin aspart, supporting improved post-meal glycaemic management in appropriate patients.
Dosage and Administration
Available Strength and Presentation
- 10 mL multidose vial
- Concentration: 100 units/mL (U-100)
- Route: Subcutaneous injection
Individualised Dosing
The total daily insulin requirement varies depending on body weight, dietary intake, physical activity, and concurrent medical conditions. Dosing must be tailored based on regular blood glucose monitoring and clinical assessment.
Timing of Injection
Fiasp should be injected:
- Up to 2 minutes before starting a meal, or
- Within 20 minutes after beginning a meal
Use in Type 1 Diabetes
In Type 1 diabetes, Fiasp is typically incorporated into a basal–bolus insulin regimen. Approximately 50% of the total daily insulin requirement is commonly delivered as rapid-acting mealtime insulin, with the remainder supplied by a longer-acting basal insulin.
Use in Type 2 Diabetes
In Type 2 diabetes, Fiasp may be added when oral antidiabetic medicines alone do not achieve adequate glycaemic control. It may be used alongside basal insulin and/or other glucose-lowering agents.
Patients should receive structured education on injection technique, dose adjustment, and hypoglycaemia recognition in accordance with NHS best practice recommendations (see NHS guidance on insulin therapy).
Clinical Benefits of Rapid-Acting Insulin Aspart
- Fast onset of action to address post-meal blood glucose spikes
- Flexible dosing window around meals
- Suitable for both adult and paediatric patients
- Compatible with basal insulin regimens
Effective postprandial glucose management contributes to improved overall HbA1c control and may reduce the risk of long-term diabetes-related complications when used appropriately as part of a comprehensive treatment strategy.
Possible Side Effects
Common Adverse Effects
- Hypoglycaemia (low blood sugar)
- Injection site reactions (redness, swelling, itching)
- Allergic skin reactions such as rash
Serious Adverse Reactions
Severe hypoglycaemia is the most significant risk associated with insulin therapy. Symptoms may include confusion, sweating, tremor, palpitations, seizures, or loss of consciousness. Immediate treatment with fast-acting carbohydrates is essential, and urgent medical attention should be sought in cases of severe or persistent hypoglycaemia.
Patients and caregivers should be trained to recognise early warning signs and follow their prescribed hypoglycaemia management plan.
Drug Interactions
Insulin requirements may be altered by concomitant medications. Examples include:
- Oral antidiabetic agents
- ACE inhibitors
- Diuretics
- Beta-blockers (which may mask hypoglycaemia symptoms)
- Thiazolidinediones (TZDs)
Patients should inform their prescriber of all prescription medicines, over-the-counter products, and herbal supplements prior to initiating or adjusting insulin therapy.
Precautions and Important Considerations
- Do not use during episodes of hypoglycaemia
- Use with caution in renal or hepatic impairment
- Inform your clinician of any insulin allergies
- Discuss use during pregnancy or breastfeeding
- Monitor blood glucose closely during illness or stress
Adjustments to insulin dose may be required in cases of kidney or liver disease due to altered insulin metabolism. Careful monitoring is essential during pregnancy, and treatment decisions should follow specialist medical advice.
Storage Instructions
Before First Use
- Store in a refrigerator at 2°C to 8°C
- Do not freeze
- Keep in the original carton to protect from light
After Opening
- May be kept at room temperature below 30°C
- Use within 28 days
- Protect from excessive heat and direct sunlight
Discard any vial that has been frozen or exposed to extreme temperatures. Keep all medicines out of sight and reach of children.
Buying Fiasp Online Safely
Purchasing insulin online may offer convenience and potential cost savings. Patients should ensure that any pharmacy supplying prescription medicines operates legally and requires a valid prescription. Always consult your healthcare professional before sourcing medicines internationally to confirm suitability and regulatory compliance.
For additional guidance, refer to the General Pharmaceutical Council (GPhC) register to verify UK pharmacy registration.
Frequently Asked Questions (FAQs)
What type of insulin is Fiasp?
Fiasp is a rapid-acting insulin analogue containing insulin aspart, formulated for mealtime glucose control.
When should Fiasp be injected?
It should be administered up to 2 minutes before eating or within 20 minutes after starting a meal, as directed by a healthcare professional.
Can Fiasp be used in children?
Yes, Fiasp may be prescribed for paediatric patients with diabetes under specialist supervision.
Is Fiasp suitable for Type 2 diabetes?
Yes, it may be used in adults with Type 2 diabetes when insulin therapy is required to achieve glycaemic targets.
Reviews
There are no reviews yet.