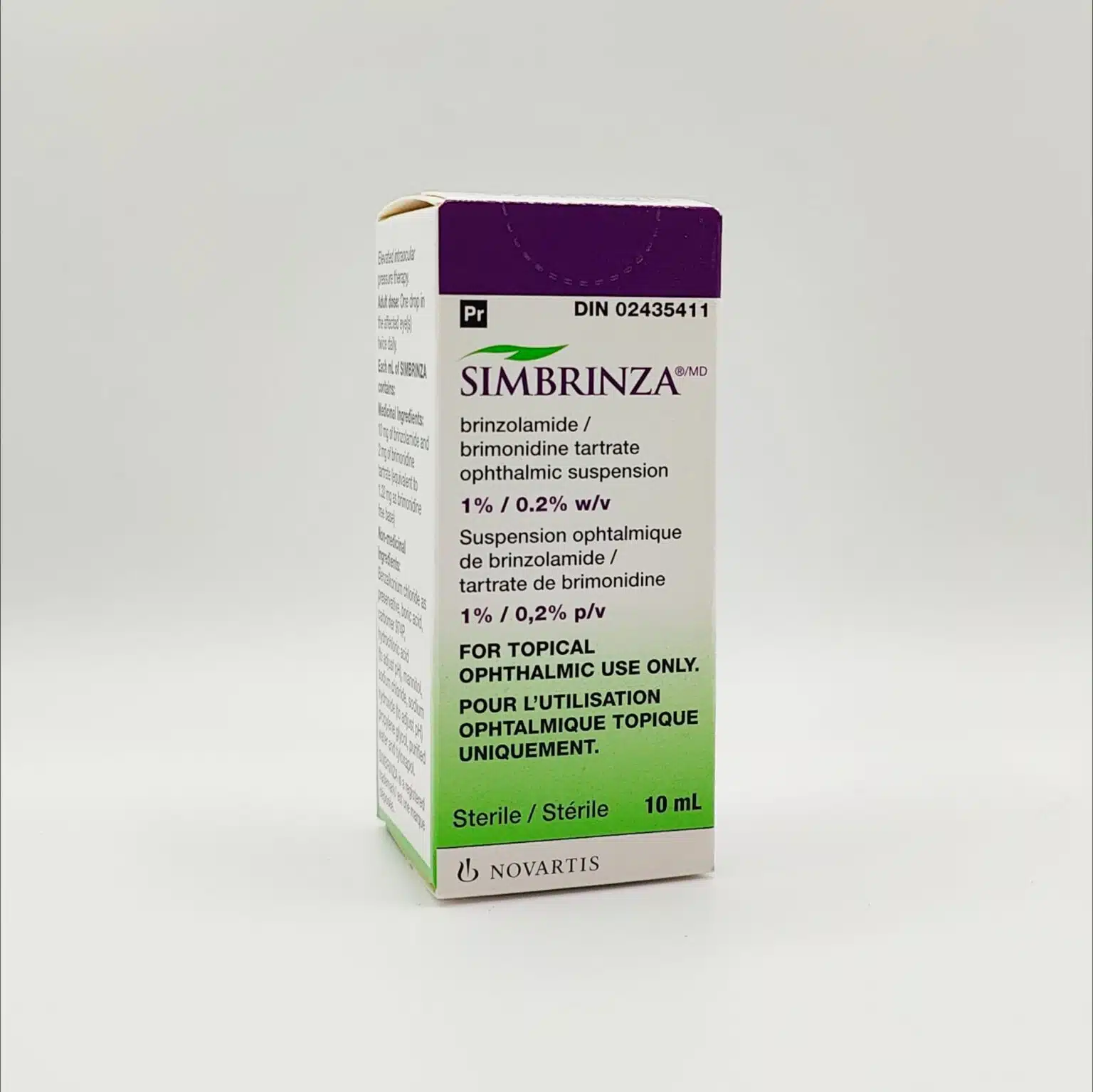
Simbrinza (Brinzolamide/Brimonidine Tartrate) Ophthalmic Suspension
Simbrinza is a prescription-only ophthalmic suspension combining brinzolamide and brimonidine tartrate to reduce elevated intraocular pressure (IOP) in adults with open-angle glaucoma or ocular hypertension. Its dual-action formula lowers aqueous humour production and enhances fluid outflow to help protect the optic nerve and preserve vision.
Overview of Simbrinza Eye Drops
Simbrinza is a fixed-dose combination eye drop indicated for the reduction of elevated intraocular pressure (IOP) in patients diagnosed with open-angle glaucoma or ocular hypertension, particularly where monotherapy has not achieved sufficient pressure control. Persistent elevation of IOP is the primary modifiable risk factor for optic nerve damage and progressive visual field loss in glaucoma.
The formulation contains two pharmacologically distinct active ingredients: brinzolamide, a carbonic anhydrase inhibitor, and brimonidine tartrate, an alpha-2 adrenergic receptor agonist. By targeting different mechanisms involved in aqueous humour dynamics, Simbrinza offers enhanced IOP reduction compared with single-agent therapy.
According to the NICE guideline on glaucoma diagnosis and management, effective IOP control is central to slowing disease progression. Combination ophthalmic preparations such as Simbrinza may improve adherence by simplifying treatment regimens in appropriate patients.
What Is Simbrinza Used For?
- Reduction of elevated intraocular pressure in open-angle glaucoma
- Management of ocular hypertension
- Patients requiring additional IOP lowering beyond single-agent treatment
Simbrinza is not indicated for acute angle-closure glaucoma. Any sudden onset of severe eye pain, blurred vision, halos around lights, or nausea requires urgent ophthalmic assessment.
How Simbrinza Works: Dual Mechanism of Action
Brinzolamide – Carbonic Anhydrase Inhibition
Brinzolamide inhibits carbonic anhydrase II within the ciliary processes of the eye. This enzyme is involved in the formation of bicarbonate ions, which are essential for aqueous humour secretion. By reducing bicarbonate production, brinzolamide decreases fluid formation inside the eye, thereby lowering intraocular pressure.
Brimonidine Tartrate – Alpha-2 Adrenergic Agonism
Brimonidine tartrate stimulates alpha-2 adrenergic receptors in the eye. This action both reduces aqueous humour production and increases uveoscleral outflow. The dual influence on fluid dynamics results in a clinically meaningful reduction in IOP.
Complementary Therapeutic Effect
By combining two agents with distinct yet complementary actions, Simbrinza provides additive pressure-lowering benefits. Fixed-combination therapy may also reduce exposure to preservatives compared with administering separate bottles, which may be advantageous in patients with ocular surface sensitivity.
The NHS overview of glaucoma emphasises that consistent daily treatment is critical to maintaining stable eye pressure and protecting the optic nerve from irreversible damage.
Simbrinza Dosage and Administration
The usual recommended dosage is one drop in the affected eye(s) twice daily. Your prescribing clinician may adjust the regimen based on individual response and tolerance.
Important Administration Guidance
- Shake the bottle thoroughly before each use.
- Wash hands prior to application.
- Avoid allowing the dropper tip to touch the eye or surrounding surfaces.
- If using additional ophthalmic medications, wait at least 5 minutes between drops.
- Discard the bottle 125 days after opening.
Step-by-Step Application Instructions
- Tilt your head back slightly.
- Gently pull down the lower eyelid to create a small pocket.
- Instil one drop into the conjunctival sac.
- Close your eyes gently.
- Apply nasolacrimal occlusion by pressing lightly at the inner corner of the eye for 1–2 minutes to reduce systemic absorption.
Nasolacrimal occlusion is particularly important with brimonidine to minimise systemic exposure and reduce potential side effects such as drowsiness or low blood pressure.
How To Use Simbrinza Safely
Do not discontinue treatment without medical advice. Glaucoma is typically a chronic condition requiring long-term management. Abrupt cessation may result in uncontrolled intraocular pressure.
If you miss a dose, apply the next dose at the scheduled time. Do not double-dose to compensate for a missed application.
Possible Side Effects of Simbrinza
Common Side Effects
- Blurred vision immediately after instillation
- Dry mouth
- Fatigue or drowsiness
- Ocular irritation or redness
Less Common but Serious Side Effects
- Allergic reactions including swelling of the face or throat
- Difficulty breathing
- Severe ocular inflammation
Seek immediate medical attention if you experience symptoms of an allergic reaction or breathing difficulty. Patients should report persistent eye pain, discharge, or significant visual changes promptly.
Drug Interactions
Simbrinza may interact with certain systemic medications. Inform your healthcare provider if you are taking:
- Monoamine oxidase inhibitors (MAOIs)
- Tricyclic antidepressants
- Other alpha-adrenergic agonists
- Oral carbonic anhydrase inhibitors
- Antihypertensive medications
Concomitant use with MAOIs is contraindicated due to the potential for enhanced systemic effects of brimonidine. Always provide a full medication history before initiating therapy.
Precautions and Contraindications
Before using Simbrinza, inform your doctor if you:
- Have renal or hepatic impairment
- Have a history of sulfonamide allergy (brinzolamide is a sulfonamide derivative)
- Are pregnant, planning pregnancy, or breastfeeding
- Are taking antidepressant medications
Simbrinza is not recommended in children or in patients with severe renal impairment. It should not be used in patients currently receiving MAOI therapy or in individuals with narrow-angle glaucoma.
Patients may experience temporary blurred vision after instillation. Avoid driving or operating machinery until clear vision is restored.
Buying Simbrinza Online
Purchasing Simbrinza through a licensed pharmacy such as LifePath Meds may provide cost savings compared with local retail prices. However, safety and authenticity must remain the priority.
Guidelines for Safe Online Purchasing
- Verify licensing: Ensure the pharmacy is properly regulated and requires a valid prescription.
- Consult your healthcare provider: Confirm that international purchase complies with your treatment plan.
- Understand import regulations: The U.S. FDA generally permits importation of up to a 90-day personal supply.
Avoid websites that dispense prescription medications without a prescription, as counterfeit or substandard products pose serious health risks.
Storage Instructions for Simbrinza
Store Simbrinza at room temperature between 2°C and 25°C (36°F to 77°F). Keep the bottle tightly closed and protect from excessive light and moisture.
Discard the bottle 125 days after first opening, even if some solution remains. Keep out of reach of children and pets.
Frequently Asked Questions (FAQs)
What is Simbrinza used for?
Simbrinza is used to lower elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension.
How often should Simbrinza be used?
The usual dosage is one drop in the affected eye(s) twice daily, as prescribed by your doctor.
Can Simbrinza be used with other glaucoma drops?
Yes, but a minimum interval of five minutes should separate different ophthalmic medications to prevent washout effects.
Where can I access the official prescribing information?
The full product monograph is available here: Simbrinza Official Product Monograph.


Reviews
There are no reviews yet.