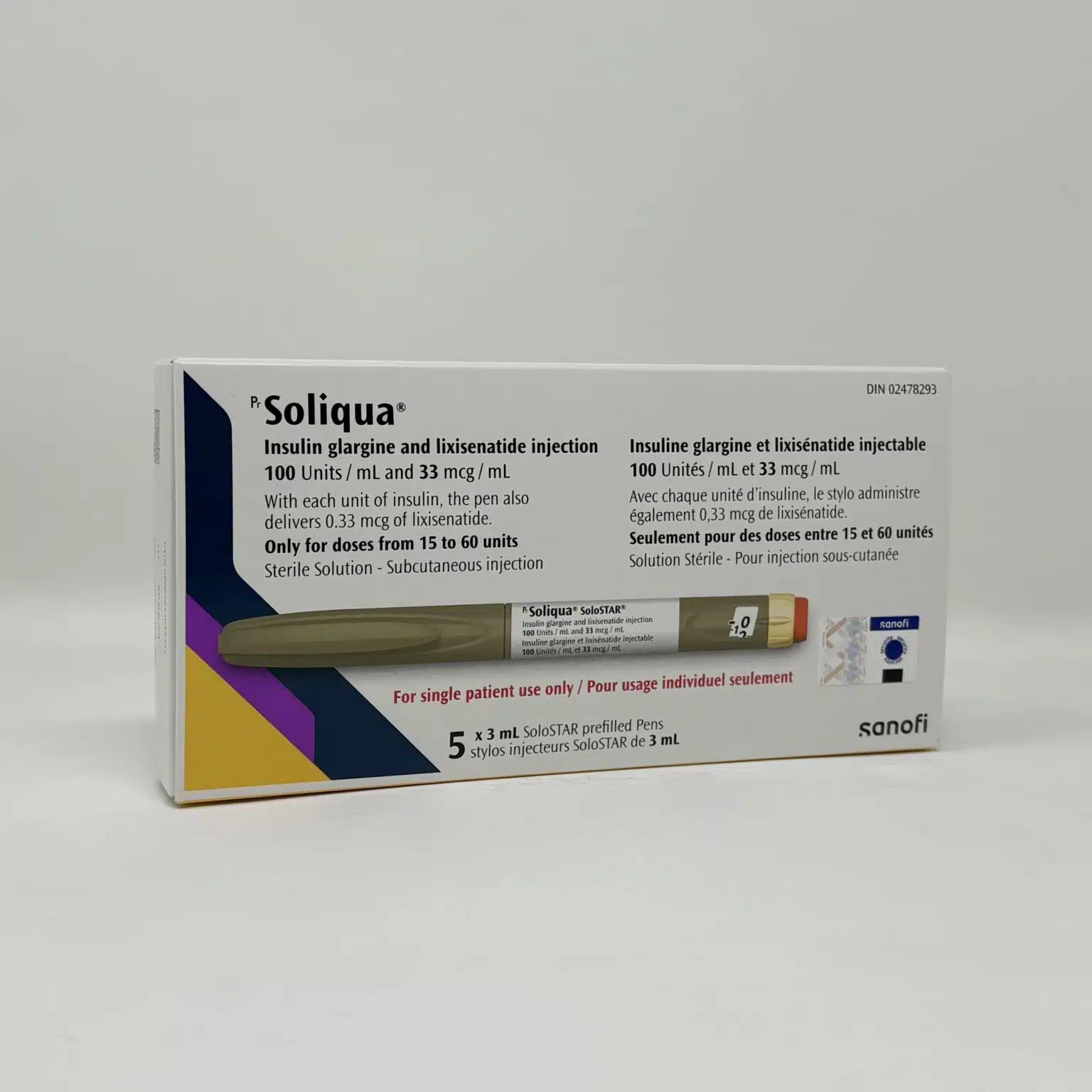
Soliqua SoloStar Pen 100/33 (Insulin Glargine / Lixisenatide)
Soliqua SoloStar 100/33 is a once-daily combination injectable treatment for adults with type 2 diabetes mellitus who require improved glycaemic control despite basal insulin therapy. It contains insulin glargine (a long-acting basal insulin analogue) and lixisenatide (a GLP-1 receptor agonist), working together to regulate fasting and postprandial blood glucose levels in a single prefilled pen device.
Comprehensive Overview of Soliqua SoloStar for Type 2 Diabetes
Soliqua SoloStar 100/33 is a fixed-ratio combination of insulin glargine and lixisenatide, designed to simplify treatment intensification in adults with type 2 diabetes mellitus (T2DM). By integrating basal insulin with an incretin-based therapy, Soliqua addresses two key contributors to hyperglycaemia: elevated fasting glucose and excessive post-meal glucose excursions.
According to guidance from the National Institute for Health and Care Excellence (NICE) – Type 2 Diabetes in Adults, combination injectable strategies may be considered when glycaemic targets are not achieved with oral agents and basal insulin alone. Soliqua provides such an option in a single daily injection.
For patients concerned about treatment affordability, regulated online pharmacy services such as LifePath Meds may offer access to brand-name therapies at competitive prices, subject to prescription and eligibility requirements.
Therapeutic Indication
Soliqua is indicated as an adjunct to diet and exercise to improve glycaemic control in adults with type 2 diabetes mellitus who are inadequately controlled on:
- Basal insulin (less than 60 units daily),
- Or basal insulin in combination with metformin.
It is not indicated for:
- Type 1 diabetes mellitus
- Diabetic ketoacidosis
- Use alongside other GLP-1 receptor agonists
- Combination with short-acting (prandial) insulin
Treatment decisions should always align with current clinical guidelines and individual patient assessment.
How Soliqua Works: Dual-Mechanism Glucose Control
1. Insulin Glargine – Basal Insulin Coverage
Insulin glargine is a long-acting insulin analogue that provides relatively steady insulin activity over approximately 24 hours. It lowers fasting plasma glucose by facilitating cellular glucose uptake and suppressing hepatic glucose production.
2. Lixisenatide – GLP-1 Receptor Agonist Action
Lixisenatide is a glucagon-like peptide-1 (GLP-1) receptor agonist. It works through several complementary mechanisms:
- Enhances glucose-dependent insulin secretion
- Suppresses glucagon release when glucose levels are elevated
- Slows gastric emptying, reducing postprandial glucose spikes
By combining these mechanisms in one pen, Soliqua addresses both basal and post-meal hyperglycaemia, potentially improving glycaemic control while simplifying injection regimens.
Composition, Strength and Pen Specifications
Soliqua SoloStar is supplied as a prefilled, disposable pen for subcutaneous injection.
- 100 units insulin glargine per mL
- 33 micrograms lixisenatide per mL
Each 3 mL pen contains:
- 300 units of insulin glargine
- 100 micrograms of lixisenatide
One unit of Soliqua delivers 1 unit of insulin glargine and 0.33 micrograms of lixisenatide. The maximum daily dose is 60 units.
Dosing and Administration Guidelines
Administration Timing
Soliqua should be administered once daily within one hour before the first meal of the day. Inject subcutaneously into the abdomen, thigh, or upper arm, rotating sites with each injection to reduce the risk of lipodystrophy.
Initial Dosing
- Patients previously using <30 units basal insulin: Start at 15 units once daily.
- Patients previously using 30–60 units basal insulin: Start at 30 units once daily.
Patients requiring fewer than 15 units or more than 60 units daily should use alternative therapeutic options.
Titration
Dose adjustments are typically made in increments of 2–4 units weekly, based on fasting blood glucose readings and clinical response. Close glucose monitoring is recommended during initiation and adjustment phases.
Missed Dose Advice
If a dose is missed, administer within one hour prior to the next meal. Do not double the dose to compensate for a missed injection.
Common Adverse Effects
Frequently reported side effects include:
- Hypoglycaemia (low blood glucose)
- Nausea
- Vomiting
- Diarrhoea
- Headache
- Nasopharyngitis or upper respiratory tract infections
- Injection site reactions
Gastrointestinal effects are generally attributable to lixisenatide and are often transient.
Hypoglycaemia Risk and Management
Hypoglycaemia is the most common adverse reaction associated with insulin-containing regimens. Symptoms may include sweating, tremor, palpitations, confusion, dizziness, and headache.
Severe hypoglycaemia may result in seizures or loss of consciousness and requires urgent medical attention. Patients are advised to carry fast-acting carbohydrates (e.g., glucose tablets) at all times.
Serious Warnings and Precautions
- Personal or family history of medullary thyroid carcinoma
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Hypersensitivity to insulin glargine or lixisenatide
Additional precautions include:
- Potential risk of pancreatitis
- Possible renal impairment, particularly with dehydration
- Delayed gastric emptying affecting absorption of oral medicines
- Potential increases in heart rate and PR interval prolongation
Soliqua must never be administered intravenously or intramuscularly and should not be mixed with other insulin preparations.
Drug Interactions
Medicines that influence blood glucose may necessitate dose adjustments. Oral medicines requiring rapid absorption should be taken at least one hour before injection. Oral contraceptives should be taken either one hour before or eleven hours after Soliqua administration.
Alcohol, corticosteroids, beta-blockers, and certain psychiatric or cardiovascular medicines may alter glycaemic control.
Comparison with Other Diabetes Therapies
Compared with basal insulin alone, Soliqua offers additional postprandial control via GLP-1 receptor agonist activity. In contrast to separate insulin and GLP-1 injections, it provides a simplified once-daily regimen that may support treatment adherence.
However, it may not be appropriate for individuals requiring flexible mealtime insulin or very low/high insulin doses.
Storage and Handling
- Store unused pens refrigerated at 2°C–8°C.
- Do not freeze.
- Protect from light.
- After first use, follow manufacturer guidance regarding room temperature storage duration.
Access and Affordability
The cost of combination injectable diabetes therapies can be substantial. Purchasing through regulated pharmacy referral services such as LifePath Meds may reduce out-of-pocket expenses while maintaining access to authorised products. Always ensure compliance with local regulations and prescription requirements.
Reviews
There are no reviews yet.